Article Information
Ronaldo Santiago1*, Jak Ozsarfati1, Holly Shulman1, Reuben Valenzuela2, Michael Zitney1
1. Meditech Rehabilitation Centre, ON, Canada
2. HSHS medical group, Neuroscience center, IL, United States
*Corresponding Author: Ronaldo Santiago, Meditech Rehabilitation Centre, Toronto, ON, Canada, Tel: 4162511055
Received: 14 December 2021; Accepted: 21 December 2021; Published: 03 January 2022
Abstract
Background: Several studies have detailed the efficacy of Photobiomodulation Therapy (PBMT) as a treatment for concussions [1]. As there are no widely accepted imaging or laboratory modalities that document concussive injuries, monitoring improvement objectively proves difficult. ImPACT® is a computer based neurocognitive assessment tool meant to measure cognitive performance and subjective symptoms for concussion injuries. This study uses ImPACT® as a means to document an improvement in symptomatology for patients with persistent post-concussion symptoms pre and post-treatment with PBMT [2].
Methods: This retrospective study reviews patient performance in the ImPACT® test in a cohort of 35 patients who are diagnosed with mild Traumatic Brain Injuries (mTBI) and experiencing persistent post-concussive symptoms. Patients initially took the Post Injury 1 test then underwent PBMT therapy using the BIOFLEX DUO+ system three times a week for 4 weeks using approved parameters for treatment of the cervical spine soft tissue injuries. After the 4 week treatment, patients took the Post Injury 2 test and the results were compared.
Results: All patients improved clinically after PBMT and their Post injury 2 ImPACT® test results were significantly better compared to their Post injury 1 ImPACT® test results.
Conclusion: PBMT may provide neurological rehabilitation in patients with persistent post-concussion symptoms and ImPACT® is an appropriate and objective measure to monitor and assess improvement.
Keywords
Photobiomodulation Therapy, Post-Concussion Syndrome, Mild Traumatic Brain Injury, Neuro-Cognitive Assessment
Article Details
1. Introduction
Photobiomodulation therapy (PBMT) therapy is a well-established modality for treating many musculoskeletal conditions. Several groups are currently considering and investigating PBMT as a treatment for several different neurological disorders [1]. This study reports the use of PBMT as a treatment modality for post-concussive symptoms. We used the Immediate Post-Concussion Assessment and Cognitive Testing (ImPACT®) tool to objectively assess patients’ improvement in cognition and symptomatology. Concussion, also called mild traumatic brain injury (mTBI), is an acute neuro-physiological event related to traumatically induced blunt impact applied to the head and/or neck due to sudden acceleration, deceleration or rotational forces with a transient disturbance of brain function [3, 4].MTBI often cause neurological, psychiatric and cognitive problems. Typically, patients with mTBI suffer from headache, fatigue, dizziness, cognitive and memory impairment, depression, emotional outbursts/mood lability, impaired judgment, impul-sivity, loss of executive skills, neck pain and sensitivity to environmental stimuli (light, sound, computer screens) [5-8].The Centers for Disease Control and Prevention (CDC) estimated that about 2.87 million TBI-related emergency department (ED) visits, hospitalizations, and deaths occurred in 2014 [9]. The current estimates of the sports-related concussions and brain injuries in the US are 1.6–3.8 million every year [10].The annual percent change in Canada was reported by Rao et al as 9.6 (95% CI 8.2– 11.0) for all ages, 10.3 (95% CI 2.5–18.1) for youth and 9.7 (95% CI 9.4–10.0) for adults between 2005 and 2014, which showed an increased incidence of TBIs.According to the same study, the proportion of Canadians who reported having a TBI in 2014 more than doubled to 3.2%, compared to 1.4% in 2005. There were approximately 155,000 TBI cases in 2014 in Canada [11].
Although most symptoms resolve in 1 to 3 weeks, approximately 5–43% of concussed individuals have experienced persistent symptoms lasting weeks or months [12, 13]. Post-concussion syndrome (PCS) was a catch-all term for the various persistent post-concussive symptoms and was previously accepted as a valid diagnosis. It was defined as the persistence of physical, cognitive, emotional, and sleep symptoms beyond the usual recovery period of 7 to 21 days after a concussion [14]. Both the World Health Organization (WHO), and the Diagnostic and Statistical Manual of Mental Disorders (DSM) no longer contain a specific entry for post-concussion syndrome. Instead, the 11th revision of the Interna-tional Classification of Diseases (ICD-11) of the WHO captures persistent post-concussive symptoms under mild neurocognitive disorder, defined as a “subjective experience of a decline from a previous level of cognitive functioning, accompanied by objective evidence of impairment in performance on one or more cognitive domains” [15] that may be attributable to several conditions, including trauma. The fifth edition of the DSM likewise reclassified post-concussion syndrome as major or mild neuro-cognitive disorder due to traumatic brain injury. The diagnostic criteria has specified that the neuro-cognitive disorder must persist beyond the “acute post-injury period” [16].
1.1 Background and rationale
There has been no definitive treatment for persistent post-concussive symptoms. Previous suggested treatments focused primarily on alleviating individual symptoms and included antidepressants, antihypert-ensive and/or anti-epileptic agents, focused (inclu-ding vestibular) rehabilitation, occupational therapy, psychotherapy and sleep therapy [17]. This has led to interest in other healing alternatives such as PBMT.
1.2 Photobiomodulation therapy
The concept of PBMT, also called Low Level Laser Therapy, in its present form as a treatment for various disorders has been around since the 1960s. Cyto-chrome c oxidase (CCO) is a terminal enzyme of the electron transport chain that mediates the electron transfer from cytochrome c to molecular oxygen and therefore influences the mitochondrial membrane potential and the consequent processes that follow such as Adenosine Triphosphate (ATP) production and the release of Nitric Oxide (NO) [18-21]. Various studies suggest that pigments associated with CCO act as photoreceptors and responsive to a specific wavelength window in the electromagnetic spectrum, concentrated specifically within the visible red light ( ∽660 nm) and invisible near infrared light (NIR) wavelengths ( ∽810 nm). Within these wavelengths, light stimulation leads to increased production of ATP and the cascade of processes that accompanies this, from tissue repair, decreased inflammation and vasodilation. The effect of light, and PBMT specifically, on the mitochondria and at the cellular level is the basis for the wide application of PBMT in various disorders.
All traumatic brain injuries, regardless of the severity, can cause an increase in cerebral glucose uptake, a reduction of ATP levels and prolonged cerebral metabolic rate of glucose consumption (CMRglc) depression [22]. This is accompanied by a decrease in cerebral blood flow (CBF) which can remain decreased for extended durations. Maugans et al. studied 12 children ages 11 to 15 who had a concussion secondary to a sports-related injury and showed that CBF, as measured by phase-contrast magnetic resonance angiography, was decreased relative to controls immediately after injury and that this decrease persisted beyond 30 days after injury even after symptoms resolved [23]. Neural tissues contain large amounts of mitochondrial CCO. Red and NIR photonic energy modulates reactive oxygen species, activates mitochondrial DNA replication, increases early-response genes, increases growth factor expression, induces cell proliferation, and alters nitric oxide levels [24-26]. Nitric oxide (released locally) increases regional cerebral blood flow [27]. Wu et al. demonstrated that a single application of 800–810 nm NIR light within four hours of injury resulted in a considerable improve-ment in neurological function [28].
Many studies have shown neuroprotective effects of PBMT in many neurological disorders [29-33] and some studies showed the benefits of red and NIR light applications in vivo in animals with exper-imental TBI [34-36]. Initial clinical studies of PBMT for persistent TBI symptoms in humans have been encouraging. For example, Naeser et al. investigated 11 chronic TBI cases with non-penetrating brain injury and showed that these patients had significant improvements with PBMT [37]. Naeser et al. also reported on two patients with chronic TBI whose executive function and verbal memory were improved by midline and bilateral scalp application of red/NIR LED therapy [38].
1.3 ImPACT® (Immediate Post-Concussion Assessment and Cognitive Testing)
In competitive sports, as well as in rehabilitation, pain and neurological clinics, computer-based neuro-cognitive testing has become a principal component in the evaluation of concussion patients [39-42]. The most recognized computer-based neurocognitive assessment tool in North America is the Immediate Post-Concussion Assessment and Cognitive Testing (ImPACT®) (ImPACT Applications Inc, Pittsburgh, Pennsylvania) [43, 44]. ImPACT® test has been shown to be both reliable and valid for baseline and post-concussion assessment of neurocognitive fun-ction [45-47]. Meditech Rehabilitation Centre began implementing the ImPACT® test, specifically the Post-Injury test series, as a means of objectively assessing functional cognitive and symptom improve-ment in patients post-concussion.
The ImPACT® test included six tests/modules (i.e., word discrimination, design memory, x’s and o’s, symbol match, color match, three letters) that yielded five composite/domain scores (i.e., verbal memory, visual memory, visual motor skills, reaction time, and impulse control). For the verbal memory, visual memory, visual-motor skills, and reaction time composite scores, demographically adjusted percen-tile scores (i.e., age and sex) were provided on the standard clinical report printout. Demographically adjusted percentile scores for the impulse control composite score were computed from the normative data tables available from the test publisher [48]. The Cognitive Efficiency Index (CEI) is a measure tabulated from the Symbol Match section of the ImPACT® test. It was calculated based on the speed and accuracy of items clicked correctly. However, it should not be considered an overall concussion severity index nor has it been validated for that purpose. The ImPACT® test has removed the CEI in its newest streamlined version 4 format released in 2021 and has replaced it with the Two Factor score.48The ImPACT® post-concussion symptom rating scale contained 22 somatic, affective, and cognitive symptoms, with each symptom being rated from 0 (none) to 6 (severe). Two values can be computed for the symptom questionnaire: total symptom score (sum of all ratings for all symptoms) and the number of symptoms endorsed as being present (regardless of the severity). The goal of this study is to review the effect of PBMT alone as a potential treatment modality for post-concussive symptoms secondary to a mTBI and using subjective reporting and objective testing with the ImPACT® test to assess improvement in cognition and symptomatology in patients.
2. Methodology
In this retrospective study, patient files from January to December 2018 from a single clinical site in Toronto, Ontario, Canada (Meditech Rehabilitation Centre) who agreed to undergo off-label PBMT specifically for persistent post-concussive symptoms were reviewed after receiving independent IRB approval. Patients included were between the ages of 15-65 years clinically diagnosed with a qualifying mTBI within at least 3 months of the traumatic incident with persistent post-concussion symptom-matology and not currently undergoing any treatment. For reference, the clinical site used the previous International Classification of Diseases, Tenth Revision (ICD-10) clinical criteria for PCS defined as a “history of TBI and the presence of three or more of the following eight symptoms: 1) headache, 2) dizziness, 3) fatigue, 4) irritability, 5) insomnia, 6) concentration or 7) memory difficulty, and 8) intolerance of stress, emotion, or alcohol” [49] prior to the release of ICD-11 one year later. Patients who had any positive findings on cranial imaging studies, a diagnosis of or a family history of neuropsychiatric co-morbidity or any additional diagnoses compounding or complicating the diagnosis of a concussion or mTBI were excluded. As well, patients were excluded if they were undergoing other types of managed therapy with the exception of over the counter pain relievers.
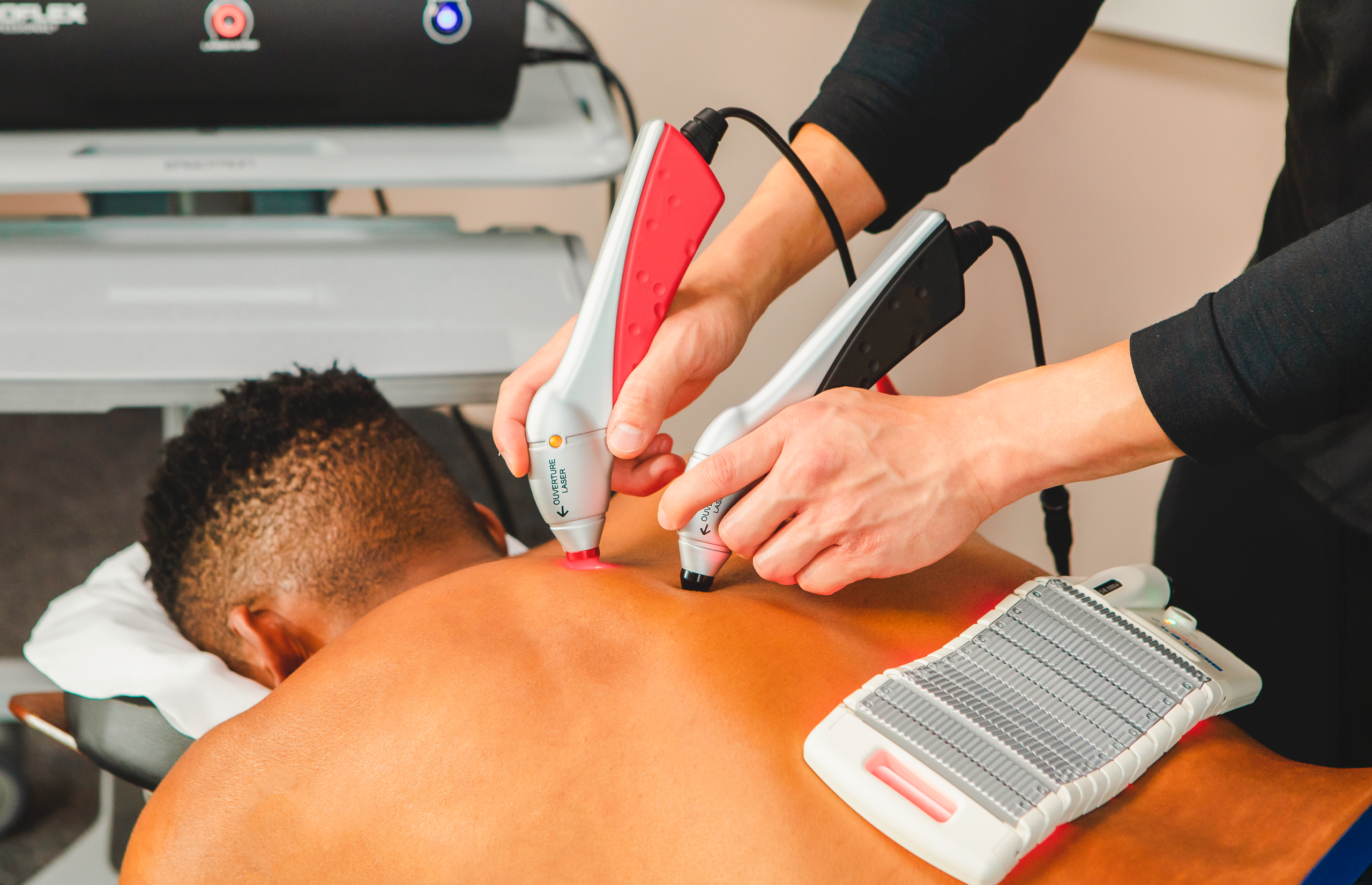
Before treatment, a thorough medical history and physical examination was completed by a primary care physician to confirm the diagnosis of PCS and the Post Injury Test 1 program of the Impact® test system was administered. After testing, patients underwent PBMT using the Bioflex® DUO 180+ Therapist system that uses a 180 Light Emitting Diode (LED) array set followed by laser probes (Red AlGaInP Laser max power at 100mW; Infrared GaAlAs Laser max power at 200mW). Both delivery methods were applied using four arranged placements to the cervical spine and six placements to the cranial area (three placements each side) simultaneously powered by two Bioflex® Mobile Professional Units, and both entailed the use of red light at 660 nm wavelength and near-infrared light at 825-840 nm wavelength. The Bioflex® DUO 180+ Therapist system is a Health Canada approved Class III Medical Device indicated for soft tissue and sports injuries, repetitive stress injuries, arthritic conditions, tissue repair & wound healing. Treatment provided is considered off-label per standard practices adopted by the clinical site for patients presenting with PCS seeking alternative therapy. Placements are described in image 1. Treatment was provided three times per week on alternating days with weekends off for four weeks for a total of 12 treatments using a variable dosing scale wherein increasing total power dosage and modulation is applied every week per the schedule adopted in Table 1. Patients are treated and tested in the same darkened room free of noise interruptions. At the end of the 12 weeks, all patients took a second ImPACT® test (Post Injury 2 test) and the results were compared with Post Injury 1.
|
Week 1 |
|||||||
|
Treatment Head Wavelength |
Frequency (Hz 5 μs resolution) |
Duty Cycle (10% steps) |
Duration (sec) |
Average Power Output (mW) |
Power Density (mW/cm2) |
Energy (J) |
Energy Density (J/cm2) |
|
Static LED Arrays* |
|||||||
|
660 nm |
CW*** |
300 |
750 |
10 |
225 |
3 |
|
|
840 nm |
20 |
40 |
360 |
600 |
8 |
216 |
2.88 |
|
Kinetic Spot Laser Diodes** |
|||||||
|
660 nm (100% power) |
CW*** |
7 |
75 |
750 |
0.525 |
5.25 |
|
|
Week 2 |
|||||||
|
Treatment Head Wavelength |
Frequency (Hz 5 μs resolution) |
Duty Cycle (10% steps) |
Duration (sec) |
Average Power Output (mW) |
Power Density (mW/cm2) |
Energy (J) |
Energy Density (J/cm2) |
|
Static LED Arrays* |
|||||||
|
660 nm |
CW*** |
300 |
750 |
10 |
225 |
3 |
|
|
840 nm |
50 |
50 |
360 |
750 |
10 |
270 |
3.6 |
|
Kinetic Spot Laser Diodes** |
|||||||
|
660 nm (100% power) |
CW*** |
7 |
75 |
750 |
0.525 |
5.25 |
|
|
825 nm (75% power) |
CW*** |
7 |
135 |
1350 |
0.945 |
9.45 |
|
|
Week 3 |
|||||||
|
Treatment Head Wavelength |
Frequency (Hz 5 μs resolution) |
Duty Cycle (10% steps) |
Duration (sec) |
Average Power Output (mW) |
Power Density (mW/cm2) |
Energy (J) |
Energy Density (J/cm2) |
|
Static LED Arrays* |
|||||||
|
660 nm |
CW*** |
300 |
750 |
10 |
225 |
3 |
|
|
840 nm |
50 |
70 |
360 |
1050 |
14 |
378 |
5.04 |
|
Kinetic Spot Laser Diodes** |
|||||||
|
660 nm (100% power) |
CW*** |
7 |
75 |
750 |
0.525 |
5.25 |
|
|
825 nm (100% power) |
CW*** |
7 |
180 |
1800 |
1.26 |
12.6 |
|
|
Week 4 |
|||||||
|
Treatment Head Wavelength |
Frequency (Hz 5 μs resolution) |
Duty Cycle (10% steps) |
Duration (sec) |
Average Power Output (mW) |
Power Density (mW/cm2) |
Energy (J) |
Energy Density (J/cm2) |
|
Static LED Arrays* |
|||||||
|
660 nm |
100 |
90 |
300 |
675 |
9 |
202.5 |
2.7 |
|
840 nm |
50 |
70 |
360 |
1050 |
14 |
378 |
5.04 |
|
Kinetic Spot Laser Diodes** |
|||||||
|
660 nm (100% power) |
CW*** |
7 |
75 |
750 |
0.525 |
5.25 |
|
|
825 nm (100% power) |
CW*** |
7 |
180 |
1800 |
1.26 |
12.6 |
|
*DUO+Arrays: RED 180 LEDs, wavelength 660 nm. Spot size 75 cm2; IR (Infrared) 180 LEDs, wavelength 840 nm. Spot Size: 75 cm2
**Laser Probes (spot treatment 7 seconds per point across areas covered by arrays): RED, wavelength 660 nm. Spot Size: 0.10 cm2; IR (Infrared), wavelength 825 nm. Spot Size: 0.10 cm2
***CW – Continuous Wave Mode / Unpulsed
Table 1: PBMT dosage and treatment schedule.
3. Results
In total, there were 35 patients included in this retrospective review, who were active patients from January to December 2018. The average age for our cohort of patients was 41 years old. 49% of patients were women and 51% of patients were men. SPSS Version 25 was used to perform all statistical tests. These included descriptive statistics (mean, median, skewness, etc.), paired sample t-tests and Wilcoxon signed rank tests. Both parametric and non-parametric statistics were employed as the data collected, from the ImPACT® test, consisted of different data types. The Wilcoxon signed rank test has been used to compare health outcomes; specifically, if there has been an improvement between the first ImPACT® test and the second ImPACT® test with PBMT. The data was first exported from the ImPACT® system into Microsoft Excel. In Excel, the dataset was cleaned to ensure that each patient was only recorded once, and every variable is listed as test 1 (pre-intervention) and test 2 (post-intervention) to facilitate the statistical testing. Any extraneous and personal information that would lead to the identification of the patient was removed. A unique patient identifier was created. A paired t-test was used to evaluate the changes in the sample population scores between pre- and post-treatment intervention. The Wilcoxon signed rank test was used to calculate the difference between the pre-intervention and post-intervention scores for the same participants. Importantly, the data being normally distributed is not a condition of a Wilcoxon signed rank test, even though it is for the t-test. A Wilcoxon signed rank test can be considered the nonparametric version of a paired t-test. Thus, the two statistical tests employed were complementary to one another.
As a component of the ImPACT® test, the patient ranked their symptoms on a scale of 1 to 6. This type of nonparametric hypothesis test can demonstrate that if the alternative hypothesis is correct – there are more positive differences after treatment (α = 0.05) – then the treatment was successful in helping the patient. SPSS automatically calculates the rank for the difference in scores between test 1 (pre-intervention) and test 2 (post-intervention) for each variable being examined. The smallest difference would receive a rank of one and the largest difference would receive n. If the difference scores were tied at any point, then they would receive the same rank regardless if it was a positive or negative difference. After each of the differences has been calculated, SPSS also attaches a positive or negative sign to each rank based on the absolute difference between test 1 and test 2. The Wilcoxon signed rank test and the paired sample t-test were used to statistically compare between pre-intervention and post-intervention for all variables being evaluated. All statistically significant results have been included below.
3.1 T-test results
The Cognitive Efficiency Index (CEI) was statisti-
cally significant at the 95% confidence level, specifically 0.007. Therefore, at the 95% confidence level there is improvement between the CEI calculated at the patients’ pre-intervention treatment and post-intervention treatment.
3.2 Wilcoxon signed rank test
Table 2 has illustrated all significant measures at the 95% confidence level. The symptom score presented summary information regarding the individual’s self-reported symptom data. A higher score reflected a higher symptom total [48]. The mean for the second total symptom (25.49) score was less than the first total symptom score (41.57). Colour Match in the ImPACT® system measured the reaction time in correctly matching a word with the corresponding colour ink [46]. The average colour match results improved from pre-intervention to post-intervention and thus were significant at the 95% confidence level.
The Verbal Memory Composite Score used Word Memory Total Percent Correct, Symbol Match Total Correct Hidden, and Three Letters Percent Total Letters Correct together, by adding these three values and dividing by three. This composite score was designed to evaluate vocabulary attention span, learning and memory [48]. The Verbal Composite Score had a higher positive rank sum (453.0) than negative rank sum (108.0). The Reaction Time Composite, an average of Average Correct RT from X’s and O’s section, Symbol Match Average Correct RT Visible/3 and Color Match Average Correct RT, had a greater negative sum rank (392.50) than positive sum rank (13.5.50). The Visual Motor Speed Composite Score was designed to “evaluate visual processing, learning and memory, and visual-motor response speed” [48]. This composite score uses the Total Number Correct/4 from the X and O section of the ImPACT® Test and the Average Counted Correctly from the Three Letters section. These two scores were added together and then divided by two. The Visual Motor Speed Composite Measure had a higher positive rank sum (504.0) than negative rank sum (126.0). The Three Letters module of the ImPACT® test measured memory and eye-hand coordination speed [48]. In this section of the test, the patient was instructed to click backwards on numbers from 25 in a 5 by 5 grid and then shown three consonants. The patient was then instructed to click backwards from 25 again and then the patient was asked to recall the three consonants displayed just previous. This section is repeated 5 times.
Table 3 has demonstrated the positive and negative sum of ranks calculated as part of the Wilcoxon signed rank test. In all instances, the positive sums were greater than the negative sums at the 95% confidence level. The PBMT intervention was effective in improving patients’ scores for several of the ImPACT® test categories including: Color Match, Verbal Composite Score, Visual Composite Score and Visual Motor Speed and Reaction Time. Although the CEI has been removed from the updated Impact® version 4, given the significance of this measure at the 95% confidence level we have decided to still include the CEI in the results as a report of its utility. Aside from the objective and rapid measurement of cognitive abilities, ImPACT® included a subjective symptom reporting scale. The CDC divides mTBI symptoms into 4 categories: Thinking/Remembering, Sleep, Emotional/Mood, and Physical [50]. The patients’ symptoms decreased significantly at the 95% confidence level for patients after receiving PBMT. The Wilcoxon Signed Rank tests detailed the decrease in the total symptom score. Thus, patients were experiencing less symptoms after the intervention than without it.
|
Relationship |
Mean of Pre intervention Score ± SD |
Mean of Post Intervention Score ± SD |
Rank Sum Z |
Rank Sum P |
|
Total Symptom Score |
41.57 ± 24.188 |
25.49 ± 21.228 |
-4.473 |
0.000 |
|
Colour Match Average Correct |
0.873 ± 0.194 |
0.809 ± 0.184 |
-3.255 |
0.001 |
|
Verbal Composite Score |
80.09 ± 13.656 |
84.83 ± 11.663 |
-3.085 |
0.002 |
|
Visual Motor Speed Composite Score |
33.054 ± 8.091 |
35.681 ± 7.215 |
-3.096 |
0.002 |
|
Reaction Time Composite Score |
0.735 ± 0.181 |
0.685 ± 0.173 |
-2.410 |
0.016 |
|
Three Letters Total Sequence Correct |
3.80 ± 1.451 |
4.51 ± 1.173 |
-3.097 |
0.002 |
|
Three Letters Total Letters Correct |
12.46 ± 3.913 |
13.74 ± 3.364 |
-2.820 |
0.005 |
|
Three Letters Per Letter Correct |
0.830 ± 0.261 |
0.916 ± 0.224 |
-2.995 |
0.003 |
|
Three Letters Average Counted |
13.731 ± 4.333 |
14.971 ± 4.232 |
-2.990 |
0.003 |
|
Three Letters Average Counted Correct |
13.440 ± 4.544 |
14.834 ± 4.193 |
-3.163 |
0.002 |
Table 2: Significant Wilcoxon Signed Rank Measures from the ImPACT® test.
|
Measure |
Sum of Ranks – Negative |
Sum of Ranks – Positive |
|
Three Letters Total Sequence Correct |
21.00 |
169.00 |
|
Three Letters Total Letters Correct |
30.00 |
180.00 |
|
Three Letters Per Letter Correct |
25.00 |
185.00 |
|
Three Letters Average Counted |
132.50 |
497.50 |
|
Three Letters Average Counted Correct |
122.00 |
508.00 |
Table 3: Three Letters Sum of Ranks from the Wilcoxon Signed Rank Test.
4. Discussion
Patients with an uncomplicated mTBI do not show any evidence of intracranial pathologies such as bleeding, subdural or epidural hematoma and/or cranial fracture on standard imaging [51]. Recent studies have shown that mTBI can cause functional neuronal disruption and structural damage in humans and animals. Due to a cerebral energy imbalance, these disruptions have the potential to manifest a wide clinical spectrum, ranging from subtle cognitive deficits only detectable on neuropsychological testing to overt neurological and behavioral symptoms [14, 51]. Although full recovery is expected within three months after concussion/mTBI, there has been a small group of concussion sufferers that have experienced persistent symptoms. A number of factors can influence the rate of recovery, including the mechanism and setting for the initial injury, age, and recurrent concussion incidents in the past. A typical mTBI patient presenting at the clinical site would have had a concussion three or more months prior to initial presentation, and had already consul-ted with and treated by other healthcare providers with no relief in their symptoms. All 35 patients noted improvement in their cognitive functions and symptoms after PBMT.
PBMT has often been used ‘off label’ as a treatment modality for post-concussion symptoms secondary to mTBI. There is unfortunately a lot of variability in the market for PBMT devices, with most PBMT devices classified and licensed from general wellness devices without public health oversight to Class IV lasers. This leads to a major concern in the accept-ance of PBMT for the treatment of neurological conditions which is the use of an appropriate and effective dosage. In many cases, especially in our own clinical observations, it has been found in PBMT that more light is not necessarily therapeutically better than less light in terms of energy delivery. This biphasic dose responses (also called Arndt-Schulz law or hormesis) shows that PBMT can simultan-eously inhibit and stimulate, and the techniques and settings for consistently achieving these effects have not always been clearly stated [52]. Because of this, most experts do not advocate the enigmatic “one size fits all” dosage and prefer tailored and individualized treatment. Observations utilizing a static dosing mec-hanism in PBMT for the treatment of neurological conditions either mention no effect, or on occasion, certain uncomfortable side effects such as headaches. In this study, treatment is provided in a gradually increasing dosing fashion, first starting with a relati-vely mild dose with continuous light application before gradually increasing the energy density appli-cation (in Joules/cm2) and adding pulse modulation in weekly intervals. We observed in our clinical practice that consistent relief of persistent post-concussion symptoms are not maintained with a static maintena-nce dose, and consistently observed a plateau period, wherein patients subjectively complain of a lack of improvement in symptoms after a week or more of the same dosage even if initially they were respond-ing positively. Although effective pulse frequencies have not been fully defined, several studies have documented that lower pulse frequencies are more effective for a variety of conditions and we maintain-ned our frequencies below 100 Hz. We speculate that some patients develop a level of tolerance to treatme-nt and that this variable dosing pattern allows tissues to continuously adjust to the treatment and absorb more photons of light as treatment progresses [53].
Of note, in this study, was the application of treatment mostly in the cervical spine. Most studies that have used PBMT for the treatment of neurological conditions tended to focus treatment only in the cranial area [26, 27, 31, 32, 35, 54-57]. Since most studies show that at most cranial penetration of most PBMT devices are minimal (up to 4 cm at most using Near Infrared Lasers and significantly less with Light Emitting Diodes, based on cadaver studies) [58], including cervical treatments may be additive in cognitive improvement due to the effect of increased cerebral circulation secondary to systemic irradiation of the carotid and vertebrobasilar arteries [59] and this indirect and remote effect may actually play an important role in rehabilitation. Persistent post-concussive symptoms after mTBI has significant functional and economic implications for the patient, their relatives, school, employers, and governmental organizations. Our study has shown that PBMT could potentially provide neurological rehabilitation in patients with post-concussive symptoms as assessed using the ImPACT® test, leading to improved cogn-itive performance and an improvement in symptom-tology. These results are quite promising and with further prospective studies with bigger sample sizes, PBMT might be considered a potential treatment option for persistent post-concussive symptoms.
4.1 Limitations
There were limitations to this study. Specifically, the sample size was small, and the treatment time was limited to one month. In clinical practice, further improvement was noted beyond the one month period, and some patients had decided to continue treatment at home utilizing portable devices. Given that the consistency of use of portable devices cannot be controlled for, we decided not to include these in our study. Increasing sample sizes, extending the treatment period, and including cerebral blood flow measurement could potentially provide additional evidence of cognitive improvement utilizing PBMT.
Recommendations
Our group did not initiate an ImPACT® baseline examination for this population given that they have already suffered an injury prior to their first presentation. Doing a baseline ImPACT® test measurement under controlled conditions may have provided a better assessment of a person’s cognitive state pre-concussion and a better comparison of improvement with the Post Injury 1 and 2 tests. Initiating a study with the baseline measurements for certain concussionprone population groups, i.e., hockey players, members of the armed forces, construction workers, may prove to be a valid option for a future study.
Declarations
Ethics Approval and Consent to Participate
This study is reviewed and approved by an independent Institutional Review Board (IRB) Advarra (6100 Merriweather Drive, Suite 600 Columbia MD 21044 Pro00050608) and performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. The IRB granted a Waiver of Consent, Assent and Parental Consent for this retrospective study.
Consent for publication
Not applicable.
Availability of data and material
The datasets used and/or analysed during the current study are not publicly available due to confidentiality laws but are available from the corresponding author on reasonable request.
Competing interests
MZ is the Medical Director of Meditech Rehabil-itation Centre. RS and JO are employed full time as clinical research staff in Meditech Rehabilitation Centre. HS is a research volunteer with Meditech Rehabilitation Centre. Meditech International is the parent company of Meditech Rehabilitation Centre and manufactures BIOFLEX® DUO+ devices. The authors do not own any shares or investment in Meditech International.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Authors’ contributions
R.S., J.O., and H.S. wrote the main manuscript text. R.S. and J.O. compiled patient treatment information and test results. H.S. provided data analysis. R.V. and M.Z. provided guidance and final editing. All authors reviewed the final manuscript.
Acknowledgement
We would like to offer acknowledgement to Dr. Fred Kahn, President and CEO of Meditech International and Vyacheslav Kim, Clinic Manager of Meditech International who supervised and directed treatment.
References
- Salehpour F, Mahmoudi J, Kamari F, et al. Brain photobiomodulation therapy: a narra-tive review. Mol Neurobiol 55 (2018): 6601-6636.
- Faul M, Xu L, Wald MM, et al. Traumatic Brain Injury in the United States: Emergency Department Visits, Hospitalizations and Deaths 2002-2006. Atlanta, GA: Centers for Disease Control and Prevention, National Center for Injury Prevention and Control (2010).
- Shrey DW, Griesbach GS, Giza CC. The pathophysiology of concussions in youth. Phys Med Rehabil Clin N Am 22 (2011): 577-602.
- Barkhoudarian G, Hovda DA, Giza CC. The molecular pathophysiology of concussive brain injury. Clin Sports Med 30 (2011): 33-48.
- Vaishnavi S, Rao V, Fann JR. Neuropsychiatric problems after traumatic brain injury: unraveling the silent epidemic. Psychosomatics 50 (2009): 198-205.
- Shauna Kashluba, Robin A Hanks, Joseph E Casey, et al. Neuropsychologic and functionnal outcome after complicated mild traumatic brain injury. Ach Phys Med Rehabil 89 (2008): 904-911.
- Kennedy JE, Jaffee MS, Leskin GA, et al. Posttraumatic stress disorder and posttra-umatic stress disorder-like symptoms and mild traumatic brain injury. J Rehabil Res Dev 44 (2007): 895-920.
- Polinder S, Cnossen MC, Real RGL, et al. A multidimensional approach to post-concussion symptoms in mild traumatic brain injury. Front Neurol 9 (2018): 1113.
- Centers for Disease Control and Prevention, U.S. Department of Health and Human Services. Surveillance Report of Traumatic Brain Injury-related Emergency Department Visits, Hospitalizations, and Deaths—United States (2014).
- Grant L Iverson, Brian L Brooks, Michael W Collins, et al. Tracking neurophysiological recovery following concussion in sport. Brain Inj 20 (2006): 245-252.
- Deepa P Rao, Steven McFaull, Wendy Thompson, et al. Trends in self-reported traumatic brain injury among Canadians, 2005–2014: a repeated cross-sectional analysis. CMAJ Open 5 (2017): E301-E307.
- Bazarian JJ, Wong T, Harris M, et al. Epidemiology and predictors of post-concussive syndrome after minor head injury in an emergency population. Brain Inj 13 (1999): 173-189.
- Iverson GL. Outcome from mild traumatic brain injury. Curr. Opin. Psychiatry 18 (2005): 301-317.
- Carroll LJ, Cassidy JD, Peloso PM, et al. Prognosis for mild traumatic brain injury: results of the WHO Collaborating Centre Task Force on Mild Traumatic Brain Injury. J Rehabil Med (2004): 84-105.
- World Health Organization. International statistical classification of diseases and related health problems (11th ed.) (2019).
- American Psychiatric Association. Diagnostic and statistical manual of mental disorders (5th ed.) (2021).
- Tator CH. Concussions and their conse-quences: current diagnosis, management and prevention. CMAJ 185 (2013): 975-979.
- Wang X, Tian F, Soni S, et al. Interplay between up-regulation of cytochrome-c-oxidase and hemoglobin oxygenation induced by near-infrared laser. Sci Rep 6 (2016): 30540.
- Hamblin M. Mechanisms and Mitochondrial Redox Signaling in Photobiomodulation. Photochem Photobiol 94 (2018): 199-212.
- Lanzafame R. Light Dosing and Tissue Penetration: It Is Complicated. Photobio-modul Photomed Laser Surg 38 (2020): 393-394.
- Zein R, Selting W, Hamblin M. Review of light parameters and photobiomodulation efficacy: dive into complexity. J. of Biomedical Optics 23 (2018): 120901.
- Maugans TA, Farley C, Altaye M, et al. Pediatric sports-related concussion produces cerebral blood flow alterations. Pediatrics 129 (2012): 28-37.
- Mester E, Szende B, Tota JG. Effect of laser on hair growth of mice. Kiserl Orvostud 19 (1967): 628-631.
- Chung H, Dai T, Sharma SK, et al. The nuts and bolts of low-level laser (light) therapy. Ann Biomed Eng 40 (2012): 516-533.
- Wu HM, Huang SC, Vespa P, et al. Redefining the pericontusional penumbra following traumatic brain injury: evidence of deteriorating metabolic derangements based on positron emission tomography. J Neuro-trauma 30 (2013): 352-360.
- Ando T, Xuan W, Xu T, et al. Comparison of therapeutic effects between pulsed and continuous wave 810-nm wavelength laser irradiation for traumatic brain injury in mice. PLoS One 6 (2011): e26212
- Ilic S, Leichliter S, Streeter J, et al. Effects of power densities, continuous and pulse frequencies, and number of sessions of low-level laser therapy on intact rat brain. Photomed Laser Ther 24 (2006): 458-466.
- Wu Q, Xuan W, Ando T, et al. Low-level laser therapy for closed-head traumatic brain injury in mice: effect of different wave-lengths. Lasers Surg Med 44 (2012): 218-226.
- Schiffer F, Johnston AL, Ravichandran C, et al. Psychological benefits 2 and 4 weeks after a single treatment with near infrared light to the forehead: A pilot study of 10 patients with major depression and anxiety. Behav Brain Funct 5 (2009): 46.
- Salehpour F, Rasta SH. The potential of transcranial photobiomodulation therapy for treatment of major depressive disorder. Rev Neurosci 28 (2017): 441-453.
- da Silva T, da Silva FC, Gomes AO, et al. Effect of photobiomodulation treatment in the sublingual, radial artery region, and along the spinal column in individuals with multiple sclerosis: Protocol for a randomized, controlled, double-blind, clinical trial. Medicine 97 (2018): e0627.
- Rojas JC, Bruchey AK, Gonzalez-Lima F. Low-level light therapy improves cortical metabolic capacity and memory retention. J Alzheimers Dis 32 (2012): 741-752.
- Hamblin MR. Shining light on the head: Photobiomodulation for brain disorders. BBA Clin 6 (2016): 113-124.
- Xuan W, Vatansever F, Huang L, et al. Transcranial low-level laser therapy improves neurological performance in traumatic brain injury in mice: effect of treatment repetition regimen. PLoS One 8 (2013): e53454.
- Oron A, Oron U, Streeter J, et al. Low-level laser therapy applied transcranially to mice following traumatic brain injury significantly reduces long-term neurological deficits. J Neurotrauma 24 (2007): 651-656.
- Xuan W, Agrawal T, Huang L, et al. Low-level laser therapy for traumatic brain injury in mice increases brain derived neurotrophic factor (BDNF) and synaptogenesis. J Biophotonics 8 (2015): 502-511.
- Naeser MA, Zafonte R, Krengel MH, et al. Significant improvements in cognitive performance post-transcranial, red/near-infrared light-emitting diode treatments in chronic, mild traumatic brain injury: Open-protocol study. J Neurotrauma 31 (2014): 1008-1017.
- Naeser MA, Saltmarche A, Krengel MH, et al. Improved cognitive function after transcranial, light-emitting diode treatments in chronic, traumatic brain injury: two case reports. Photomed Laser Surg 29 (2011): 351-358.
- Schatz P, Zillmer EA. Computer-based assessment of sports-related concussion. Appl Neuropsychol 10 (2003): 42-47.
- Schatz P, Browndyke J. Applications of Computer-based Neuropsychological Assess-ment. J Head Trauma Rehabil 17 (2002): 395-410.
- Vanessa C Fazio, Mark R Lovell, Jamie E Pardini, et al. The relation between post concussion symptoms and neurocognitive performance in concussed athletes. NeuroRehabilitation 22 (2007): 207-216.
- Meehan WP, d’Hemecourt P, Collins CL, et al. Computerized neurocognitive testing for the management of sport-related concussions. Pediatrics 129 (2012): 38-44.
- Randolph C, McCrea M, Barr WB. Is neuropsychological testing useful in the management of sport-related concussion?. J Athl Train 40 (2005): 139-152.
- Dessy AM, Yuk FJ, Maniya AY, et al. Review of Assessment Scales for Diagnosing and Monitoring Sports-related Concussion. Cureus 9 (2017): e1922.
- Schatz P, Sandel N. Sensitivity and specificity of the online version of ImPACT in high school and collegiate athletes. Am J Sports Med 41 (2013): 321-326.
- Elbin RJ, Schatz P, Covassin T. One-year test-retest reliability of the online version of ImPACT in high school athletes. Am J Sports Med 39 (2011): 2319-2324.
- Iverson GL, Lovell MR, Collins MW. Validity of ImPACT for examining processing speed following sports-related concussion. J Clin Exp Neuropsychol 27 (2005): 683-689.
- ImPACT® Applications, Inc. ImPACT® Version 4 Administration and Interpretation Manual (2021).
- World Health Organization (WHO). The ICD-10 Classification of Mental and Behavioural Disorders (2021).
- Centers for Disease Control and Prevention, U.S. Department of Health and Human Services. Facts about Concussion and Brain Injury Where to Get Help (2021).
- Centers for Disease Control and Prevention, National Center for Injury Prevention and Control. Report to Congress on mild traumatic brain injury in the United States: steps to prevent a serious public health (2021).
- Huang YY, Chen AC, Carroll JD, et al. Biphasic dose response in low level light therapy. Dose Response 7 (2009): 358-383.
- Khan I, Arany P. Dosimetry for photobiomodulation therapy: response to Sommers et al. Ann Transl Med 4 (2016): 208.
- Fenghua Tian, Snehal N Hase, Gonzalez-Lima F, et al. Transcranial laser stimulation improves human cerebral oxygenation. Lasers Surg Med 48 (2016): 343-349.
- Hiroshi Nawashiro, Kojiro Wada, Kanji Nakai, et al. Focal increase in cerebral blood flow after treatment with near-infrared light to the forehead in a patient in a persistent vegetative state. Photomed Laser Surg 30 (2012): 231-233.
- Naeser MA, Zafonte R, Krengel MH, et al. Significant improvements in cognitive performance post-transcranial, red/near-infrared light-emitting diode treatments in chronic, mild traumatic brain injury: Open-protocol study. J Neurotrauma 31 (2014): 1008-1017.
- Miriam Tomaz de Magalhães, Silvia Cristina Núñez, Ilka Tiemy Kato, et al. Light therapy modulates serotonin levels and blood flow in women with headache. A preliminary study. Exp Biol Med (Maywood) 241 (2016): 40-45.
- Lanzafame R. Light Dosing and Tissue Penetration: It Is Complicated. Photobio-modul Photomed Laser Surg 38 (2020): 393-394.
- Boaz Kim, Alice Brandli, John Mitrofanis, et al. Remote tissue conditioning – An emerging approach for inducing body-wide protection against diseases of ageing. Ageing Res Rev 37 (2017): 69-78.
Link to article: Click Here